The Business Research Company’s Exocrine Pancreatic Insufficiency (EPI) Therapeutics & Diagnostics Market to hit US $8.63B by 2030
LONDON, GREATER LONDON, UNITED KINGDOM, April 26, 2026 /EINPresswire.com/ — “Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics market to surpass $9 billion in 2030. In comparison, the Gastrointestinal Drugs market, which is considered as its parent market, is expected to be approximately $91 billion by 2030, with Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics to represent around 10% of the parent market. Within the broader Pharmaceuticals industry, which is expected to be $2,513 billion by 2030, the Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics market is estimated to account for nearly 0.4% of the total market value.
Which Will Be The Biggest Region In The Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market In 2030?
North America will be the largest region in the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market in 2030, valued at $3.4 billion. The market is expected to grow from $2.5 billion in 2025 at a compound annual growth rate (CAGR) of 39%. The exponential growth can be attributed to strong reimbursement frameworks supporting enzyme therapies and diagnostics, high awareness and clinical screening rates for digestive disorders, well-established healthcare infrastructure, increasing adoption of advanced pancreatic function testing, presence of leading pharmaceutical companies, and continuous innovation in gastrointestinal treatment approaches across countries such as the USA and Canada.
Which Will Be The Largest Country In The Global Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market In 2030?
The USA will be the largest country in the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market in 2030, valued at $3.1 billion. The market is expected to grow from $2.3 billion in 2025 at a compound annual growth rate (CAGR) of 36%. The exponential growth can be attributed to strong availability of approved pancreatic enzyme replacement therapies, favorable reimbursement systems, increasing clinical focus on digestive health management, expanding collaborations between pharmaceutical companies and healthcare providers, rising adoption of advanced diagnostic protocols, and well-developed healthcare infrastructure supporting long-term treatment and monitoring.
Request A Free Sample Of The Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market Report
https://www.thebusinessresearchcompany.com/sample_request?id=12992&type=smp&utm_source=EINPresswire&utm_medium=Paid&utm_campaign=Apr_PR
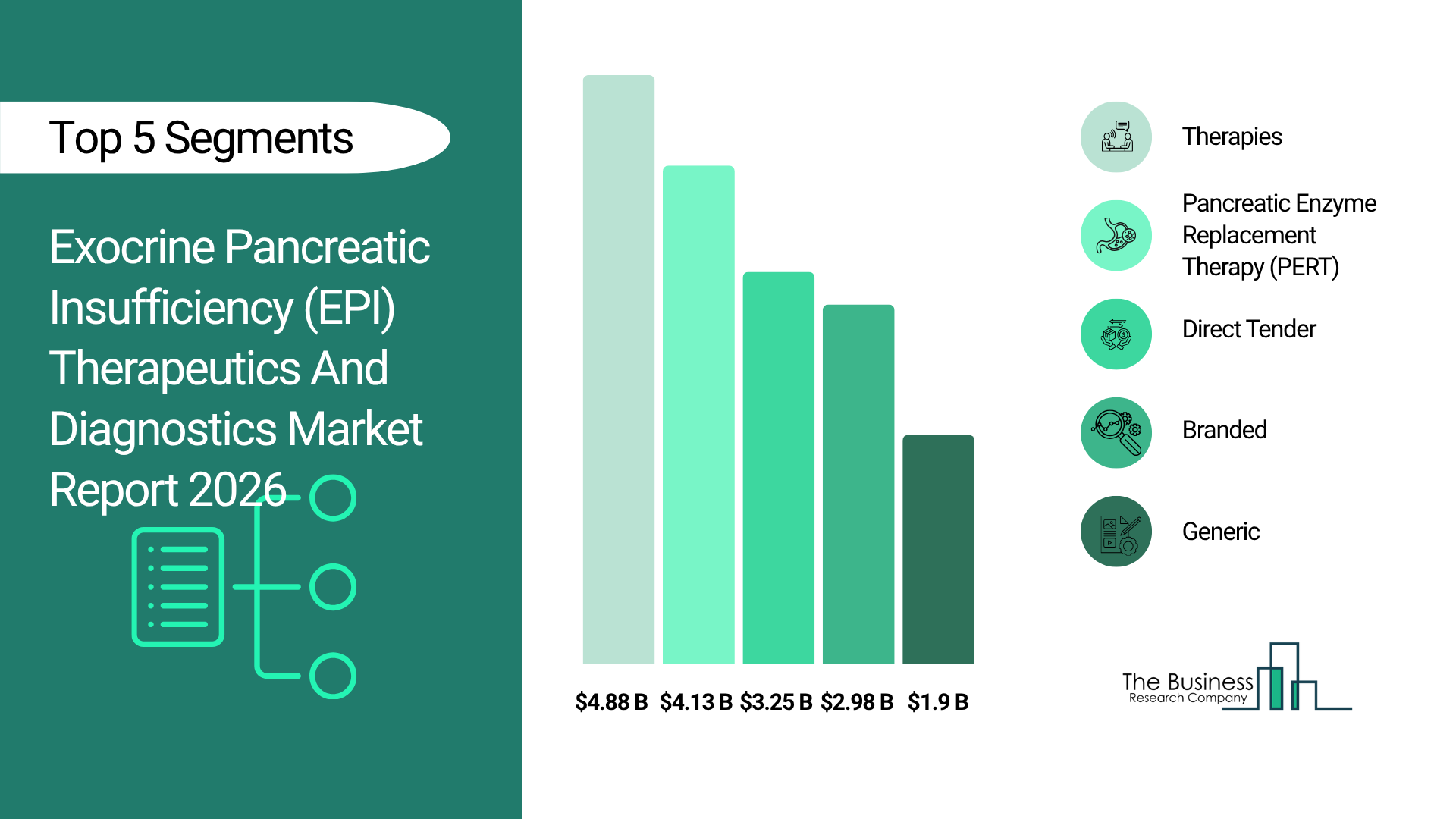
What Will Be The Largest Segment In The Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market In 2030?
The exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market is segmented by type into therapies and diagnostics. The therapies market will be the largest segment of the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market segmented by type, accounting for 74% or $6 billion of the total in 2030. The therapies market will be supported by increasing long-term dependency on enzyme supplementation for disease management, improvements in formulation stability and dosing efficiency, growing clinical guideline recommendations for early therapy initiation, rising patient adherence due to improved treatment outcomes, expanding pharmaceutical innovation in enzyme formulations, and broader access to prescription therapies across developed and emerging healthcare systems.
The exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market is segmented by distribution channel into direct tender, retail pharmacy, third-party distributor, and other distribution channels. The direct tender market will be the largest segment of the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market segmented by distribution channel, accounting for 48% or $4 billion of the total in 2030. The direct tender market will be supported by the increasing procurement of pancreatic enzyme replacement therapies and diagnostic kits by hospitals and public healthcare institutions, growing prevalence of pancreatic and gastrointestinal disorders requiring long-term treatment, rising government and institutional healthcare spending, expanding hospital-based treatment and diagnostic programs, improving access to specialty gastrointestinal medications through centralized purchasing systems, and supportive healthcare policies promoting early diagnosis and management of digestive disorders.
The exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market is segmented by end user into hospitals, specialty clinics, home care, diagnostic centers, research and academic institutes, and other end users. The specialty clinics market will be the largest segment of the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market segmented by type, accounting for 29% or $2 billion of the total in 2030. The specialty clinics market will be supported by increasing number of gastroenterology-focused clinics, rising demand for specialized and personalized digestive care, improved access to advanced diagnostic tools, growing outpatient treatment models, increasing patient preference for expert-led care, and expanding availability of targeted treatment protocols for pancreatic disorders.
What Is The Expected CAGR For The Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market Leading Up To 2030?
The expected CAGR for the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market leading up to 2030 is 7%.
What Will Be The Growth Driving Factors In The Global Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market In The Forecast Period?
The rapid growth of the global exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market leading up to 2030 will be driven by the following key factors that are expected to reshape gastrointestinal disease management, pancreatic disorder diagnosis, therapeutic innovation, and clinical treatment approaches across global healthcare systems.
Rising Prevalence Of Pancreatic Disorders And Gastrointestinal Diseases – The rising prevalence of pancreatic disorders and gastrointestinal diseases continues to significantly support the expansion of the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market by 2030. A large proportion of patients with these conditions eventually develop pancreatic enzyme deficiency, requiring long-term enzyme replacement therapy and diagnostic monitoring. As global disease prevalence rises, the patient pool needing EPI management expands significantly. This epidemiological trend strongly supports demand for both therapeutic enzymes and diagnostic tests. As a result, the rising prevalence of pancreatic disorders and gastrointestinal diseases is anticipated to contribute approximately 2.8% annual growth to the market.
Increasing Adoption Of Pancreatic Enzyme Replacement Therapy (PERT) – The increasing adoption of pancreatic enzyme replacement therapy (PERT) is expected to emerge as a major factor driving the expansion of the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market by 2030. Modern formulations containing lipase, amylase, and protease improve nutrient absorption, reduce gastrointestinal symptoms, and enhance patient quality of life. With clinical guidelines increasingly recommending early initiation of enzyme therapy, prescription rates are rising globally. Growing physician awareness and therapeutic effectiveness are therefore expanding market adoption. Consequently, increasing adoption of pancreatic enzyme replacement therapy (PERT) is projected to contribute around 2.5% annual growth to the market.
Advancements In Diagnostic Technologies And Screening Programs – The advancements in diagnostic technologies and screening programs are expected to act as a key growth catalyst for the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market by 2030. Improved diagnostic tools such as fecal elastase testing, pancreatic function tests, and genetic screening are enabling earlier and more accurate identification of EPI. Non-invasive tests with high sensitivity have significantly improved detection rates in patients with digestive disorders. Expanded screening programs and improved diagnostic infrastructure are helping identify previously undiagnosed cases. This diagnostic expansion increases treatment initiation rates and strengthens the overall market demand. Therefore, the advancements in diagnostic technologies and screening programs is projected to contribute approximately 2.0% annual growth to the market.
Access The Detailed Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market Report Here
https://www.thebusinessresearchcompany.com/report/exocrine-pancreatic-insufficiency-epi-therapeutics-and-diagnostics-global-market-report?utm_source=EINPresswire&utm_medium=Paid&utm_campaign=Apr_PR
What Are The Key Growth Opportunities In The Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market in 2030?
The most significant growth opportunities are anticipated in the therapies market and the diagnostics market. Collectively, these segments are projected to contribute over $3 billion in market value by 2030, driven by expansion into emerging healthcare markets, increasing focus on personalized treatment approaches, development of next-generation enzyme formulations, integration of advanced diagnostic technologies, and growing collaborations between pharmaceutical companies and diagnostic providers.
The therapies market is projected to grow by $2 billion, and the diagnostics market by $1 billion over the next five years from 2025 to 2030.
Learn More About The Business Research Company
The Business Research Company (www.thebusinessresearchcompany.com) is a leading market intelligence firm renowned for its expertise in company, market, and consumer research. We have published over 17,500 reports across 27 industries and 60+ geographies. Our research is powered by 1,500,000 datasets, extensive secondary research, and exclusive insights from interviews with industry leaders.
We provide continuous and custom research services, offering a range of specialized packages tailored to your needs, including Market Entry Research Package, Competitor Tracking Package, Supplier & Distributor Package and much more.
Disclaimer: Please note that the findings, conclusions and recommendations that TBRC Business Research Pvt Ltd delivers are based on information gathered in good faith from both primary and secondary sources, whose accuracy we are not always in a position to guarantee. As such TBRC Business Research Pvt Ltd can accept no liability whatever for actions taken based on any information that may subsequently prove to be incorrect. Analysis and findings included in TBRC reports and presentations are our estimates, opinions and are not intended as statements of fact or investment guidance.
Contact Us:
The Business Research Company
Americas +1 310-496-7795
Europe +44 7882 955267
Asia & Others +44 7882 955267 & +91 8897263534
Email: info@tbrc.info
Follow Us On:
LinkedIn: https://in.linkedin.com/company/the-business-research-company“
Oliver Guirdham
The Business Research Company
+44 7882 955267
info@tbrc.info
Visit us on social media:
LinkedIn
Facebook
X
Legal Disclaimer:
EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
![]()
Media gallery